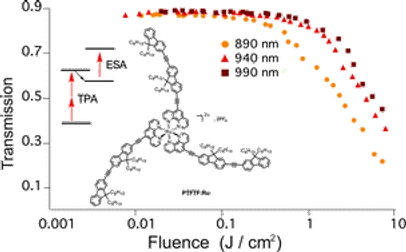
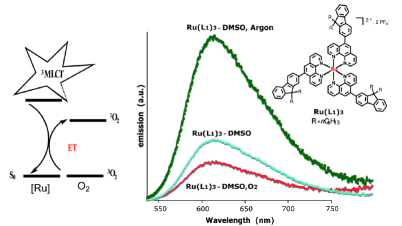
-
A modified cyclen azaxanthone ligand as a new fluorescent probe for Zn2+
H. Nouri, C. Cadiou, L.M. Lawson Daku, A. Hauser, S. Chevreux, I. Déchamps-Olivier, F. Lachaud, R. Ternane, M. Trabelsi-Ayadi, F. Chuburu and G. Lemercier
Dalton Transactions, 42 (2013), p12157-12164


DOI:10.1039/c3dt51216a | unige:29225 | Abstract | Article PDF
A new cyclen derivative L, bearing a methyl-chromeno-pyridinylidene hydrazone moiety, was synthesized and studied in MeOH, as potential fluorescent âOFF-on-ONâ sensors for Zn(II). Photocphysical properties of this ligand being PET regulated, L was only weakly emissive in the absence of metal ions (OFF). L fluorescence was increased modestly upon addition of one equivalent of Zn(II), and further increased upon addition of a second equivalent. Therefore, Zn:L behaved as a highly sensitive ON sensor for zinc. This efficiency was correlated to Zn(II) coordination via the hydrazone moiety of the fluorophore, producing an efficient CHelation-Enhanced Fluorescence (CHEF) effect. A complementary theoretical study carried out with DFT calculations further elucidated of the optical properties.